Enantiomers
Key Questions
-
Answer:
You interconvert any two groups on the chiral carbons.
Explanation:
For example, the structure of 2-brombutane is
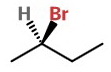
We can convert this to its enantiomer by interchanging any two groups.
We often interchange the groups on the wedges and dashes, as below.
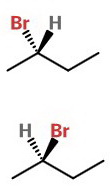
It is not immediately obvious that these are mirror images of each other.
If we rotate the top structure 180 ° about the axis and put it below our original structure, we get
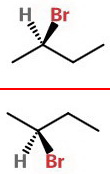
The red line represents a mirror plane that shows the enantiomers.
It is often easier to interconvert the bonds in the plane of the paper. This gives us
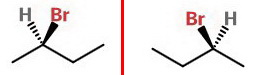
Here, the red line represents a vertical mirror plane with the two enantiomers.
There are other possibilities.
It just shows that we must practice viewing molecules in three dimensions.
-
Different configurations of molecules bind differently to the odorant receptor proteins that are part of your olfactory receptor neurons in your olfactory epithelium. Populations of these cells will "code" graded potentials in different distributions which are recognized by glomeruli and mitral neurons which send information up a pathway to your brain.
To be simple, your body can tell different enantiomers apart.