How many half-filled orbitals are in water?
1 Answer
None.
We can figure that out by looking at the MO diagram:
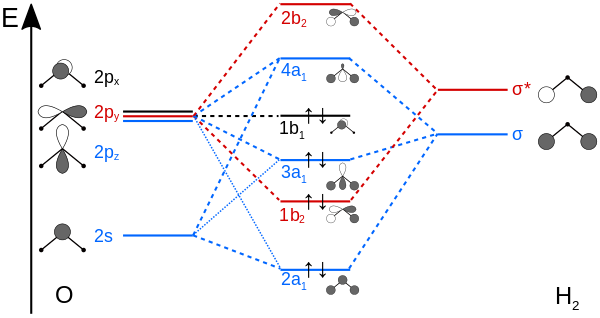
If you aren't familiar with the orbital namings (
From the Lewis structure of water:
We can say that we don't expect any half-filled orbitals in water, except if we looked at the bonding orbitals, since to the untrained eye, one might predict that shared electrons are in separate molecular orbitals (they're not, because they already overlapped).
Now let's correlate the electrons in the structure with those in the MO diagram. Note the following things we should know:
- The molecular orbital belongs more to the atom that it is closer to in energy.
- The "
#"H"_2# " in the diagram doesn't mean the molecule#"H"_2# , it means two hydrogen atoms. - Other diagrams might switch the labeling of the
#2p_x# and#2p_y# orbitals due to different conventions, so don't worry if you see things like that occurring.
Noting those things, we can conclude:
- The lone pairs of electrons are in the
#2a_1# and#1b_1# orbitals. Both belong primarily to oxygen, and thus have primarily nonbonding character. - The
#1b_2# and#3a_1# account for the bonding electrons, as they are approximately evenly shared between oxygen and each hydrogen.
Since that accounts for all the valence electrons in water, there are no half-filled orbitals at all.

