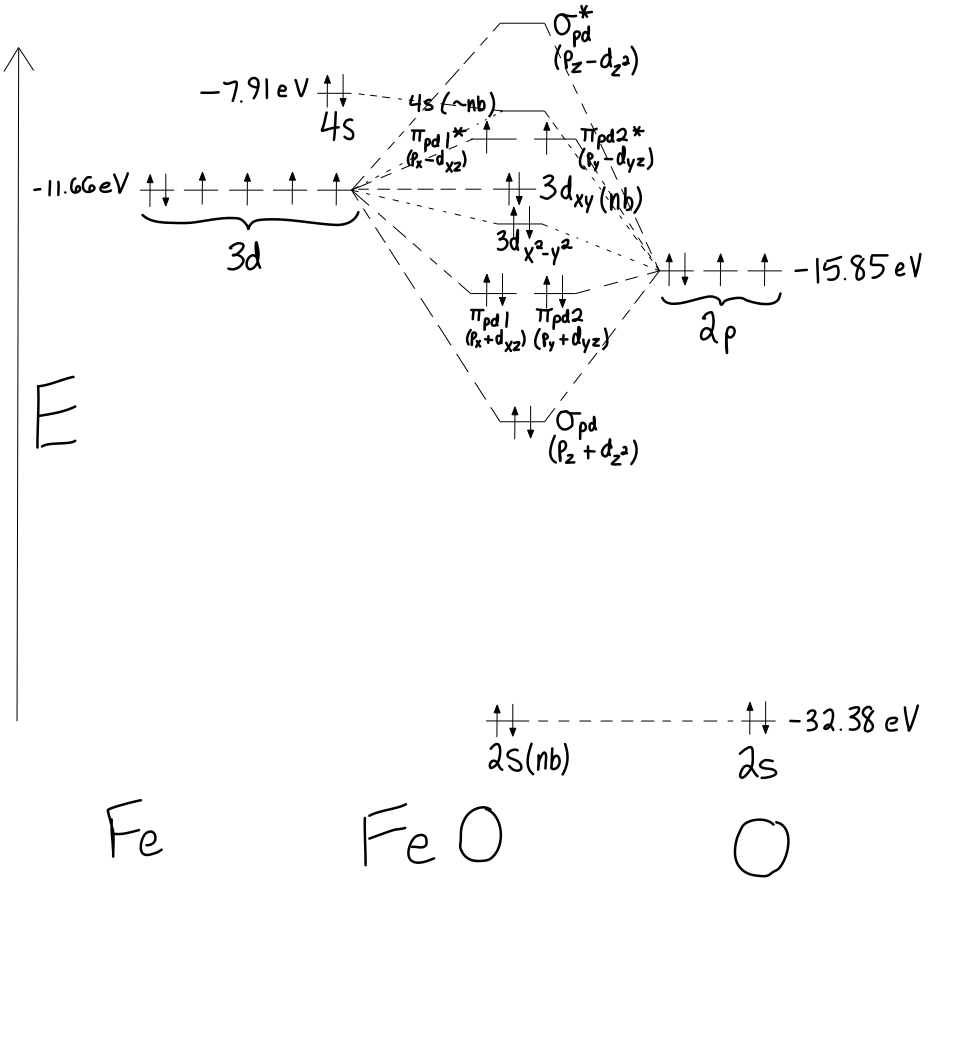
What is the MO diagram of #"FeO"#?
1 Answer
As far as I can tell, there isn't one on the internet yet... so I had to derive this one, using the orbital energies here (Appendix B.9) and knowledge of symmetry in Group Theory.
[
Here's the rundown of what we had done.
GENERAL BONDING RULES
- Conservation of orbitals must be obeyed:
There came in
#5xx3d# orbitals,#1xx4s# orbitals,#3xx2p# orbitals, and#1xx2s# orbitals, so there comes out#5 + 1 + 3 + 1 = 10# "molecular" orbitals (in this case,#"FeO"# is not really a molecule... it's an ionic compound).
- Atomic orbitals of
#(i)# significantly different energy
#(ii)# mismatched "symmetry"do not interact.
- Atomic orbitals that are
#(i)# not needed at the time (because the available compatible orbitals from the other atom have already been used up),
#(ii)# or atomic orbitals that are not interacting with the other atom's orbitals (either due to large energy differences, or symmetry mismatches)are mostly nonbonding, and at most slightly differ in energy from the respective original atomic orbitals.
- Sigma (
#sigma# ) bonds are generally going to involve greater overlap than pi (#pi# ) bonds, so#bbsigma# bonding interactions are usually going to be lower in energy than#pi# bonding interactions.
More specifically:
~ NONBONDING INTERACTIONS
- The
#2s# orbital is too low in energy (practically core-like here), so it is nonbonding. - The
#3d_(xy)# orbital of#"Fe"# does not match up with any of#"O"# atom's atomic orbital symmetries, so it is nonbonding. - The
#4s# and#3d_(x^2 - y^2)# are mostly nonbonding, because the#2p_z# orbital has already been used, and no other orbitals on#"O"# that are of that symmetry are available to pair up with these orbitals on#"Fe"# .
BONDING AND ANTIBONDING INTERACTIONS
- The bonding combinations are most likely the
#sigma_(pd)# bond from the#2p_z + 3d_(z^2)# head-on overlap, and the two#pi_(pd)# bonds from the#2p_x + 3d_(xz)# and#2p_y + 3d_(yz)# sidelong overlaps. - The antibonding combinations are therefore the
#sigma_(pd)^"*"# antibond from the#2p_z - 3d_(z^2)# head-on overlap, and the two#pi_(pd)# antibonds from the#2p_x - 3d_(xz)# and#2p_y - 3d_(yz)# sidelong overlaps.