How do you name this compound?
1 Answer
May 27, 2018
The name is (
Explanation:
The "new" (2013) IUPAC rules give chain length priority over functional groups.
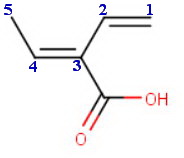
The longest chain contains five carbon atoms, so the base name is
penta-1,3-diene.
There is a
3-carboxypenta-1,3-diene.
Finally, the
The IUPAC name is (

