How is heat related to kinetic energy Like if we heat the particles, they gain kinetic energy... So,Why do the particles begin to vibrate and start their motion if we supply heat?
1 Answer
It depends on what you're heating.
- Solids have restricted degrees of freedom (they can only vibrate).
- Liquids can freely rotate and vibrate.
- Gases can freely translate, rotate, and vibrate.
As you heat, you increase the average kinetic energy of the particles.
#K_"avg" = K_("trans","avg") + K_("rot", "avg") + K_("vib", "avg")#
Depending on what kinds of motions they are capable of doing (translation, rotation, vibration), they store that energy (in accordance with their heat capacity) into that kind of energy level so that they can perform that motion.
The energy levels look like this:
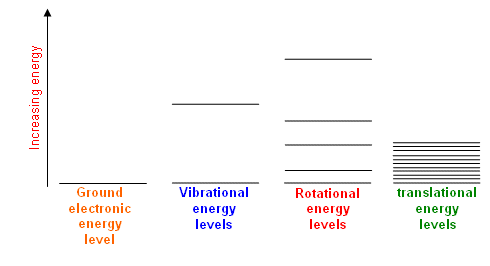
Each motion is shown below.
Translation
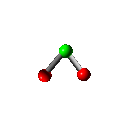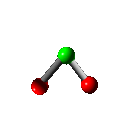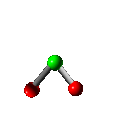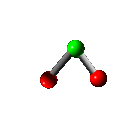
Rotation
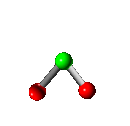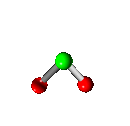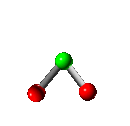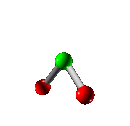
Vibration
Typically,
- Translation occurs for 100% of all molecules in the gas phase at most temperatures by default.
- Rotation occurs for 99% of all molecules in the gas phase at room temperature by default (minimum, about
#"40 K"# is needed). - Only the fundamental vibration occurs for 99% of all molecules at room temperature by default. If we want to go into excited states, we would need thousands of kelvins in temperature.
For gases, heating them immediately leads to them moving faster for precisely that reason: the translational contribution average kinetic energy vastly dominates the contributions from rotation and vibration for gases at ordinary temperatures.
Liquids don't translate faster also for that reason; instead, the energy is put into vibrating them more and rotating them more. In fact, that's what microwaves do to water; they rotate water more.
Solids can only vibrate more, but not rotate or translate.

