How is water represented in solid phase?
1 Answer
Jul 24, 2017
Ice.
Explanation:
Water, in its solid phase is known as "ice".
This occurs when the average kinetic energy of the particles in the substance decreases. As it decreases, the particles' are given an opportunity to attract other particles.
This results in a geometric pattern that the particles are arranged in.
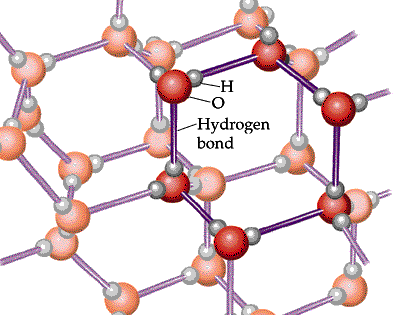
If you add energy (heat) to the substance, this would increase average kinetic energy and the particles' attraction would break free. This would turn the substance back into liquid state.
Keep on heating the substance and you'll get the gaseous state.
Hope this helps :)

