5) Sometimes secondary amines, such as pyrrolidine, are used in the alkylation of carbonyl compounds (cf. figure below). a) What problem is minimized using pyrrolidine? b) Give detailed mechanisms for all steps in the alkylation of cyclohexanone using pyr
1 Answer
May 29, 2018
Truong-Son N. answered a similar question here.
(a) What problem is minimized?
The use of pyrollidine prevents the over-reaction (the addition of more than one alkyl group) that often occurs when you use more basic reactants such as alkoxides or lithium diisopropylamide.
(b) Mechanism of alkylation
(i) Formation of the enamine
Cyclohexanone reacts with pyrrolidine in the presence of p-toluenesulfonic acid catalyst to produce 1-(1-pyrrolidinyl)cyclohexene.
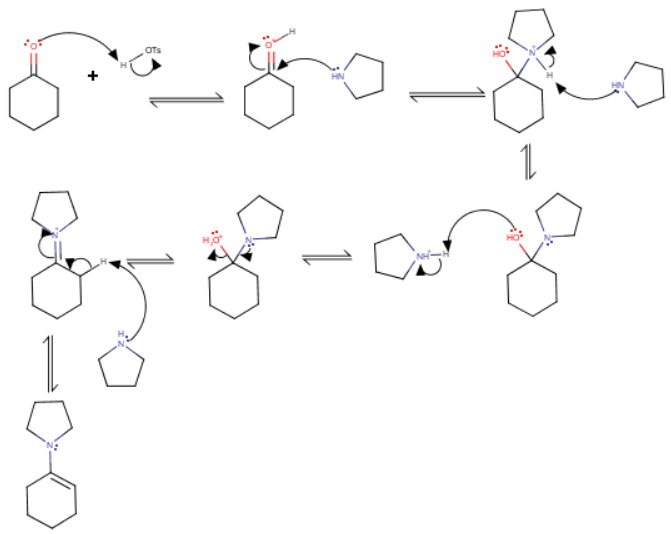
(ii) Alkylation
The enamine reacts with a primary alkyl halide, e.g. ethyl bromide to form an alkyliminium ion, which gets hydrolyzed to the ketone on workup with aqueous acid.
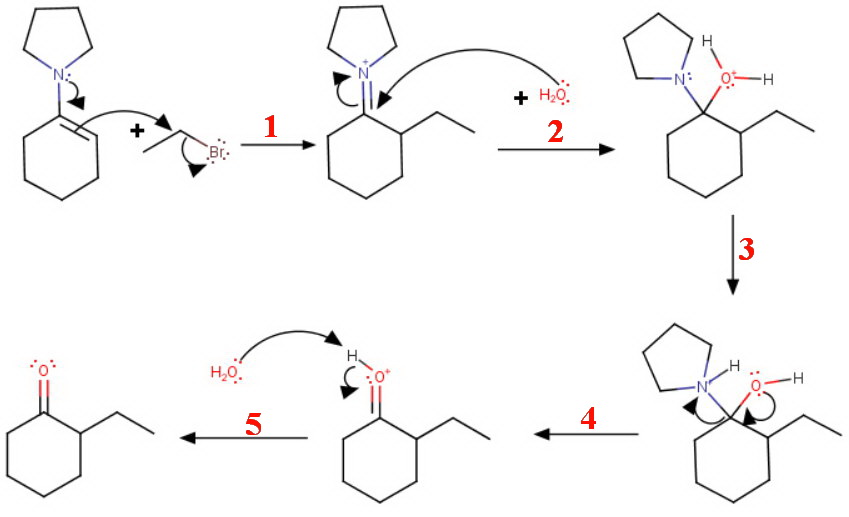
The steps in the mechanism are
#"S"_text(N)2# attack on the alkyl halide- Addition of water
- Proton transfer
- Loss of pyrrolidine
- Deprotonation

