Question #fb599
1 Answer
That's the number shown at the bottom of a box in the Periodic Table (it's also always the bigger of the two numbers).
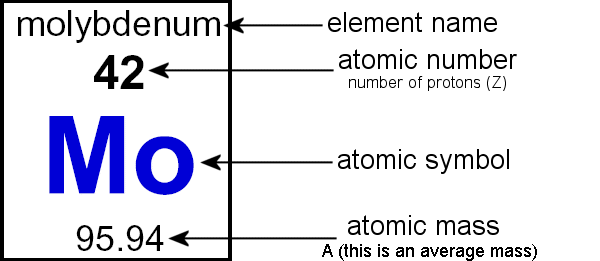
When you take the mass of an element, there are two main ways you can do it.
You can either measure the mass of one isotope of the element (isotopic mass), or you can take a sample of the element that has multiple isotopes (in the quantities they naturally occur) in it and calculate the mean average atomic mass of the sample (average atomic mass).
A good example of this is chlorine. Chlorine naturally occurs as a mixture of two isotopes: chlorine-35 and chlorine-37 (The isotopic masses of these are 35 u and 37 u respectively.).
A naturally occurring sample of chlorine is 75.78% chlorine-35 and 24.22% chlorine-37, so, to calculate the average mass, we need to do the sum
which gives us the mass shown on the periodic table, 35.5 u.
Here is a video which summarizes how to calculate average atomic mass.
video from: Noel Pauller


