Question #72084
1 Answer
The hybridization theory was put together to try and explain covalent bonding, the bonding in which electrons are shared by the two atoms.
This is not the case for ionic compounds, for which the difference in electronegativity between the two atoms (or molecules) is large enough that electrons are no longer shared, but transferred - lost and gained.
This gives rise to a postively-charged and a negatively-charged ion, which are bonded together by the electrostatic force of attraction. No hybridization is needed for these compounds since we know how ionic compounds bond - one atom loses electrons, the other one gains them.
On the other hand, hybrodization successfully explained covalent bonding. A classic example is
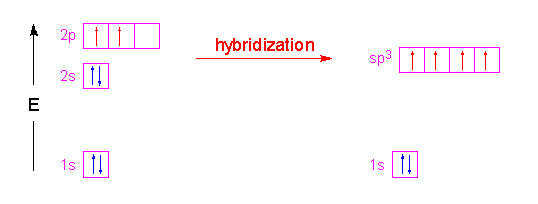
One electron is promoted from the 2s-orbital to the 2p-orbital, creating four
When it comes to the shape of an ionic compound, hybridization again cannot be used since these compounds have no definite shape.
The alternating positively-charged and negatively-charged ions which form an ionic compound arrange themselves in a tightly-packed crystal lattice structure.

This crystal lattice structure helps keep the ions tightly together, and is the reason for why ionic compounds are solid at room temperature.

