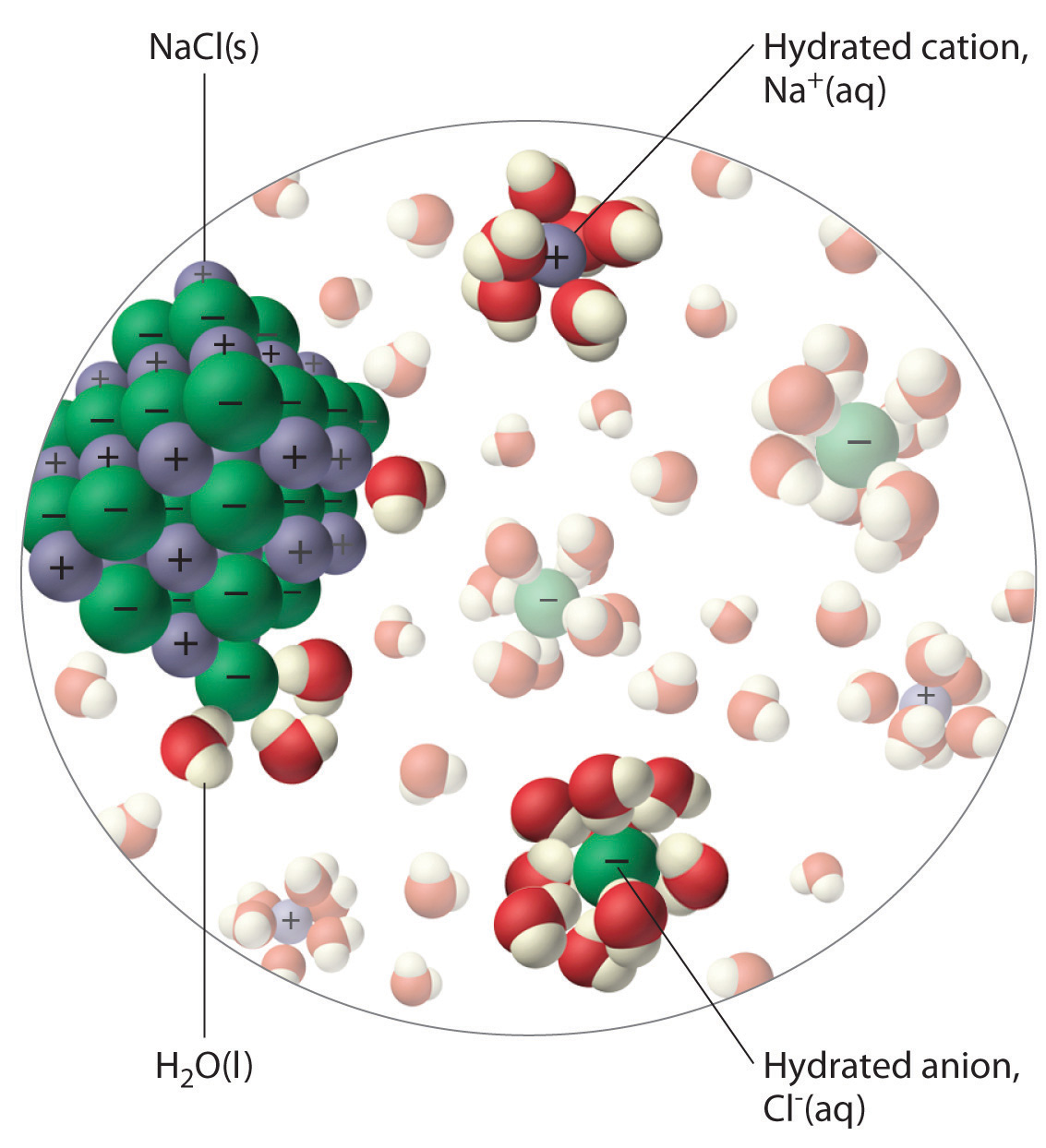
What is it called when sodium and chloride ions separate when dissolved in water?
1 Answer
This process is called dissociation. The
Sodium chloride in water forms a sodium chloride solution. Because the sodium chloride solution can conduct electricity, it is an electrolytic solution, and NaCl is an electrolyte. The following diagram illustrates the dissociation of sodium and chloride ions when dissolved in water.