Question #014c4
1 Answer
The formation of water in a fuel cell is indeed a redox reaction. Without going into too much detail, here's how a fuel cell works.
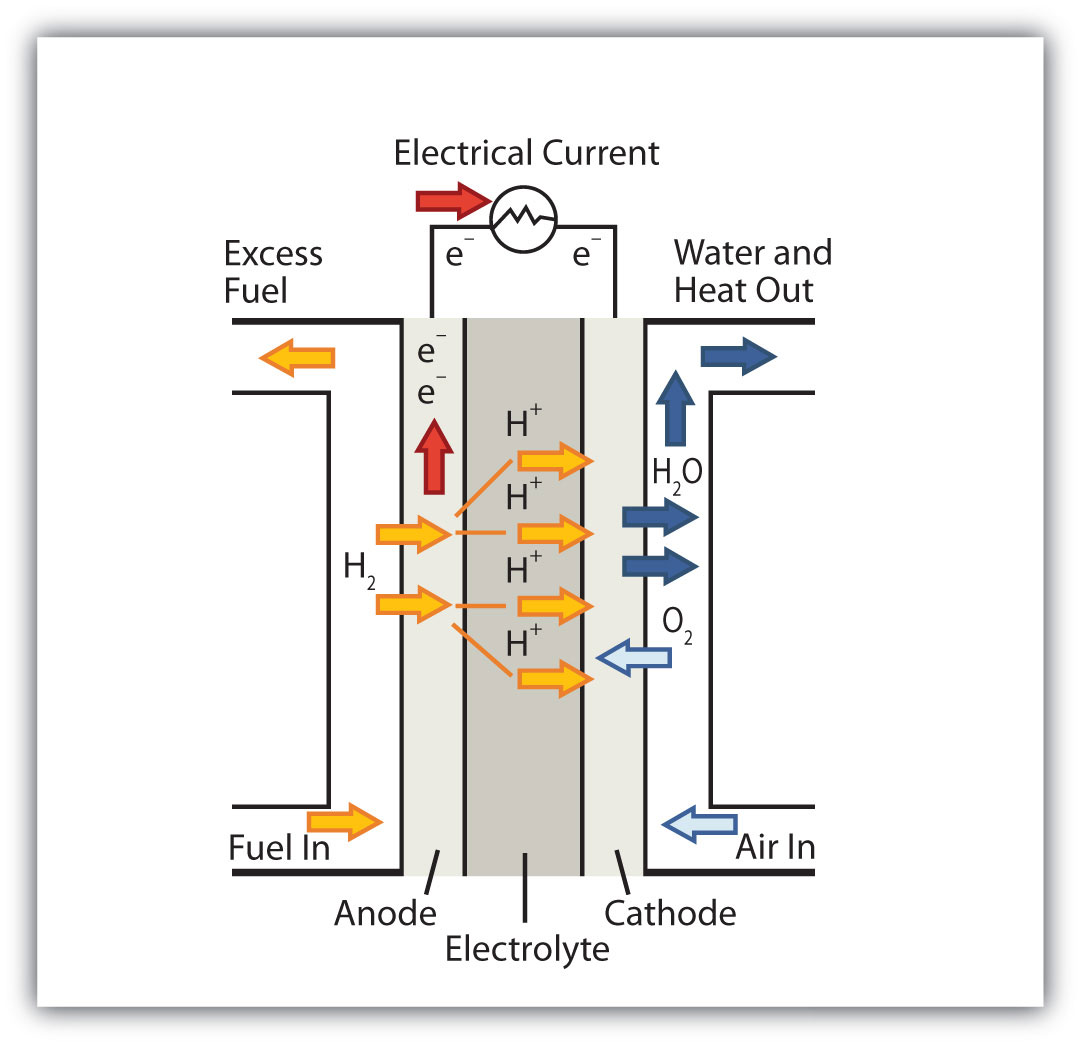
Fuel cells need hydrogen (
Hydrogen atoms are introduced at the anode and they are stripped of their electrons to form hydrogen ions. This is the oxidation reaction
The electrolyte only allows cations to pass through to the cathode, all the electrons being blocked and forced up the anode to complete the circuit.
Oxygen is introduced at the cathode and reacts with the electrons that have completed the circuit (notice that the electrons travel upwards through the anode and come down through the cathode) and with the hydrogen ions
Add these two equations to get the overall redox reaction
Since oxygen picks up two electrons from the circuit and combines with hydrogen ions, the formation of water is regarded as ionic in fuel cells.

