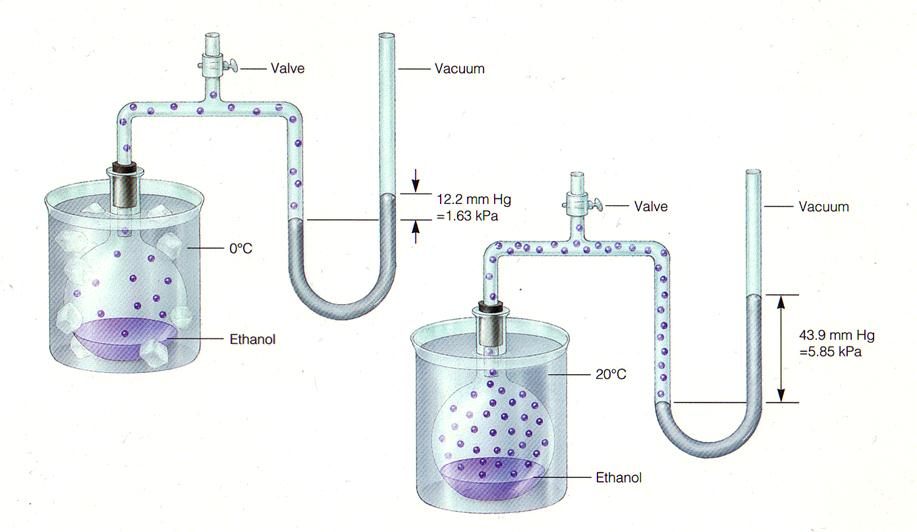
Question #2bedc
1 Answer
Vapor pressure has a direct relationship with temperature - when temperature goes up, vapor pressure increases, and when temperature goes down, vapor pressure decreases.
At the same time, vapor pressure has an inverse relationship with the strength of the intermolecular forces a particular compound has - the stronger the intermolecular forces, the lower the vapor pressure at a given temperature.
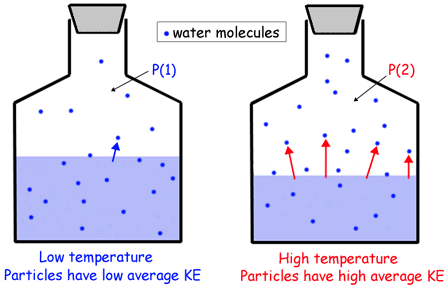
The connection between vapor pressure and temperature is made through kinetic energy, i.e. the energy of the individual molecules that make up that compound.
A higher average kinetic energy for a liquid will result in more molecules being able to escape into gas phase. Likewise, a lower average kinetic energy will result in fewer molecules being able to go into gas phase.
The bigger the number of molecules that escape into gas phase, the higher the vapor pressure will be, since now more molecules will be present above the liquid.
This concept can be seen in a plot of vapor pressure vs. temperature for water
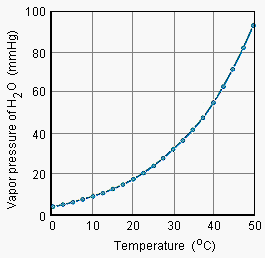
Temperature goes up
Here's how that would look from a molecular perspective

Low temperature, fewer molecules in gas phase above the liquid, lower vapor pressure.
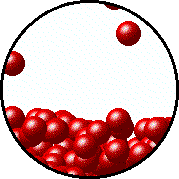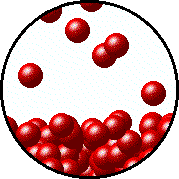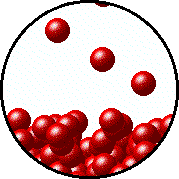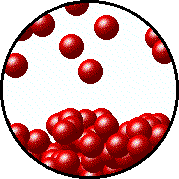
Higher temperature, more molecules in gas phase above the liquid, higher vapor pressure.