Question #8ed65
1 Answer
The solubility of potassium alum (anhydrous) is
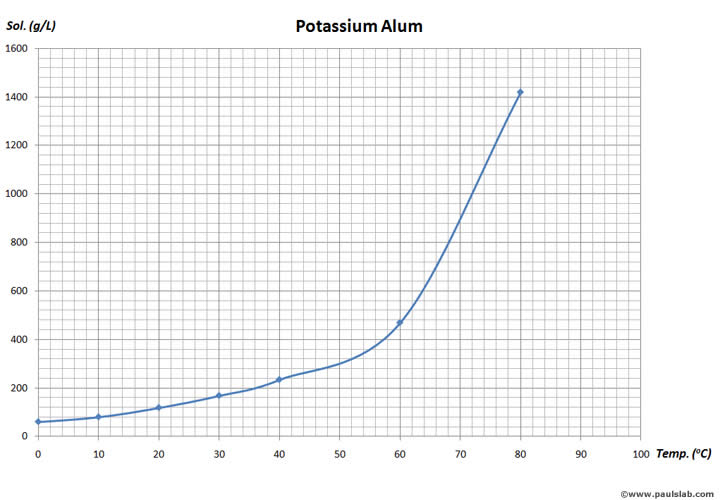
According to the above solubility chart, potassium alum's solubility exceeds
At
In the case of iron (II) oxalate, I was only able to find data on its solubility at
This very low solubility at room temperature means that iron (II) oxalate is, for all intended purposes, insoluble, or sparingly soluble at best, at this temperature.
If someone can find some reliable information on iron (II) oxalate's solubility at the requested temperature, feel free to add to the answer.

