Question #37be3
1 Answer
"For a fixed mass of gas, at a constant temperature, the product (pressure x volume) is a constant."
Pressure x Volume = constant
p x V = constant
This simply can be like this;
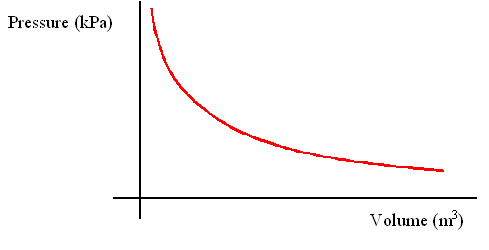
Or having x axis as 1/v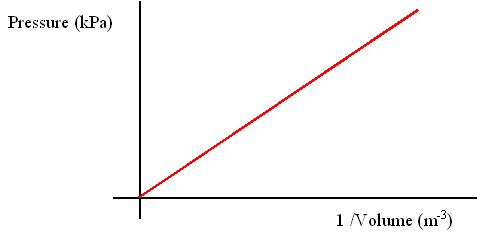
So formulating this
Hence shows the inverse relationship between pressure and volume
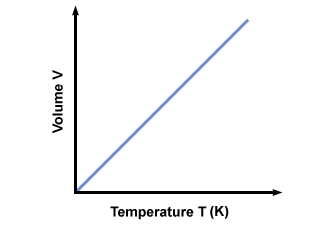
When the pressure on a sample of a dry gas is held constant, the Kelvin temperature and the volume will be directly related.
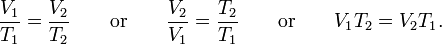
So with this in mind i would say that the pressure in your Tyre will .As temp. decreases Even the pressure decreases
Okay the graph