Question #97f87
1 Answer
Yes
Explanation:
Yes, pure water does contain ions due to a process called the autoionization (or self-ionization) of water.
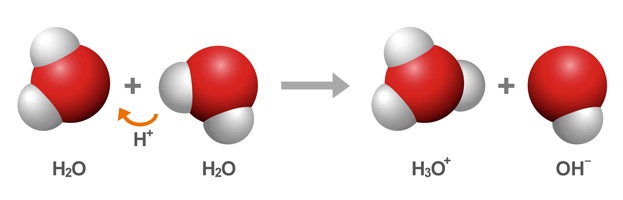
That happens because water can act both as an acid, donating a proton,
The reaction results in the formation of the hydronium ion, or
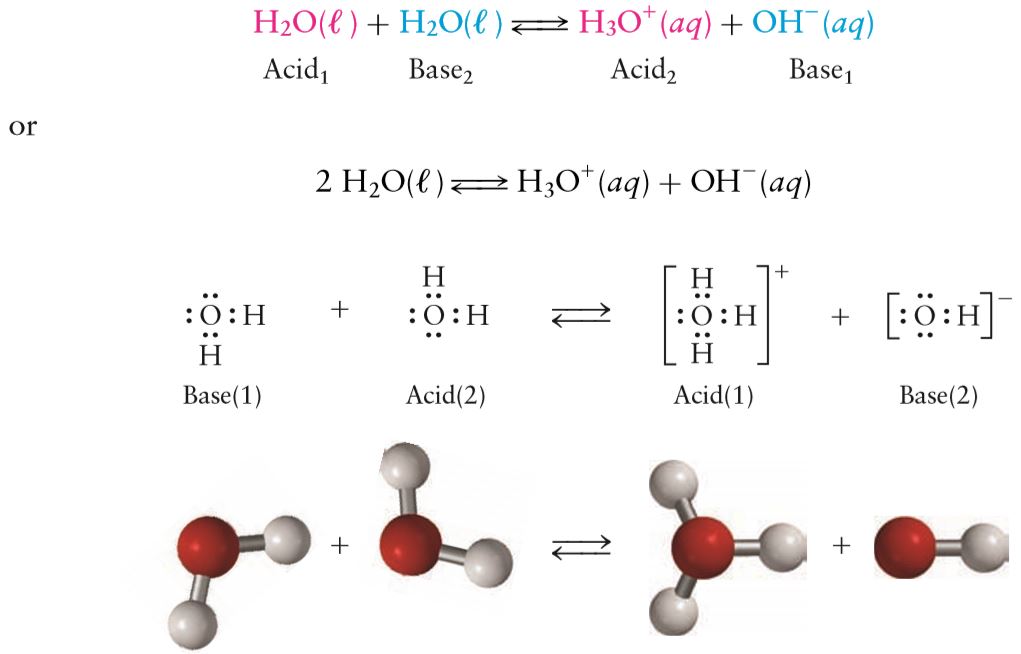
However, the concentration of hydronium and hydroxide ions will be very, very small; in fact, the equilibrium that's established in solution lies so far to the left, that only 18 in


