Question #3d17d
2 Answers
Actually, carbon monoxide does react with water.
Carbon monoxide and steam react to produce carbon dioxide,
The water gas shift reaction is a very important process used in the production of hydrogen gas for fuel cells, or in steam reforming of natural gas, a method used to produce synthesis gas.
Being an exothermic reaction, you can imagine that higher temperatures will shift the equilibrium to the left. This means that the reaction will favor the formation of the products only at relatively low temperatures.
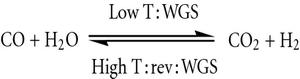
Nothing, just nothing at room temperature. But, with high temperature steam, in presence of a suitable catalyst (platinum or magnetite,


