Question #c7976
3 Answers
Simply put, because evaporation requires energy.
Explanation:
As the name suggests, evaporative cooling has something to do with evaporation and cooling.
More specifically, it describes cooling that takes place via evaporation.
The underlying principle behind this is the fact that evaporation, which is a phase change, requires energy, i.e. it is an endothermic process.
In order for the molecules that are in the liquid state to be able to go into the vapor state, they need to absorb energy.
When energy in the form of heat is absorbed by the molecules that are in liquid state, the average kinetic energy of these molecules increases. As a result, the molecules that have more kinetic energy and are closer to the surface of the liquid will escape into vapor state.
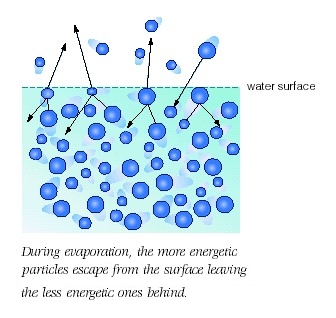
In your case, the wet bulb will become cooler as more alcohol molecules leave its surface. That happens because the energy needed to evaporate the alcohol comes from the surface of the bulb.
Think of it like this - when one molecule of alcohol evaporates from the surface, it "takes" away energy in the form of heat. This will lower the temperature of the bulb and cause the surface to feel cooler.
By comparison, the dry bulb will not experience this cooling effect because there is no evaporation taking place on its surface, which is why its temperature will be higher that that of the wet bulb.
The evaporating alcohol takes heat from the thermometer.
Explanation:
When a liquid is a a temperature below it's boiling point it still has some evaporation occurring. What is occurring on a molecular level is individual molecules (of alcohol in this case) gain enough energy to become vapor.
Now because there is no alcohol molecules in the air above the alcohol initially, it readily accepts the alcohol molecules diffusing into it. As the concentration of gaseous alcohol molecules increase, it becomes harder and harder for the liquid molecules to become gaseous because the concentration gradient becomes smaller.
Now when we add the fan, the air above the alcohol has no alcohol it in again, so the concentration gradient increases meaning it more readily accepts the gaseous alcohol.
Finally, the reason the temperature drops is because the individual molecules that gained the energy needed (those I mention in the 1st paragraph) had to get that energy from somewhere. In this case it came as heat from whatever was touching the alcohol, the glass it is sitting in, the air around in and most importantly for this experiment, the thermometer.
That is evaporative cooling.
It is sweeping away of heat from a liquid due to evaporation (or phase change) as form of latent heat.
Explanation:
The molecules in phase require more energy than liquid phase. So higher energy molecules ( molecules having higher kinetic energy) go out from the liquid lowering the liquid energy level, consequently lower temperature in the liquid.
Using of fan causes a forced evaporation or enhanced evporation in the liquid.


