Question #c8c79
1 Answer
Explanation:
The first thing you need to determine is how many atoms you get per molecule of hydrogen sulfide,
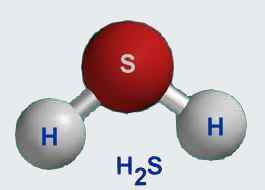
You know by looking at the subscripts in the chemical formula that one molecule of hydrogen sulfide contains
- two atoms of hydrogen
- one atom of sulfur
So one molecule contains a total of three atoms.
Next, find you how many molecules of hydrogen sulfide you get in that many moles. You know that one mole of a substance contains exactly
In your case, you will have
#0.8color(red)(cancel(color(black)("moles H"_2"S"))) * (6.022 * 10^(23)"molecules")/(1color(red)(cancel(color(black)("mole H"_2"S")))) = 4.818 * 10^(23)"molecules"#
This means that the number of atoms will be
#4.818 * 10^(23)color(red)(cancel(color(black)("molecules H"_2"S"))) * "3 atoms"/(1color(red)(cancel(color(black)("molecule H"_2"S")))) = 15.45 * 10^(23)"atoms"#
I'll leave the answer rounded to two sig figs, despite the fact that you only gave one sig fig for the number of moles of hydrogen sulfide
#"no. of atoms" = color(green)(15 * 10^(23))#

