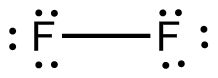Question #d2d69
1 Answer
Here's how you can do that.
Explanation:
The first thing to do here is determine the total number of valence electrons you have in a fluorine molecule,
The fluorine molecule is composed of two fluorine atoms. Each fluorine atom has
#"valence e"^(-) = 7 + 7 = 14#
Now, these two fluorine atoms have seven electrons in their outermost shells, which means that they only need one more to complete their octet.
They will thus form one single bond to which each of the two atoms contributes one electron.
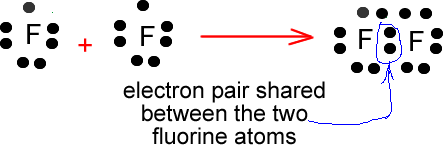
This bond will take up

SIngle bonds are drawn using a single horizontal dash between the two atoms. Lone pairs are represented using dots, each dot being equivalent to one electron.
The Lewis structure of the