Question #d16a3
1 Answer
Nov 9, 2015
Explanation:
Magnesium is in the group 2 of the periodic table that means that it's number of oxidation is 2+. Phosphate formula is
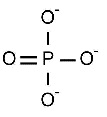
So to balance the charges Mg +2, and

