Question #85f20
1 Answer
Jan 8, 2016
Lothar Meyer's curve was a plot of atomic volume against atomic mass.
Explanation:
Meyer’s main contribution to atomic theory was his recognition of the periodic behaviour of atomic properties.
He plotted the atomic volume (the volume of a mole of atoms) of an element against its atomic mass. Here's his curve.
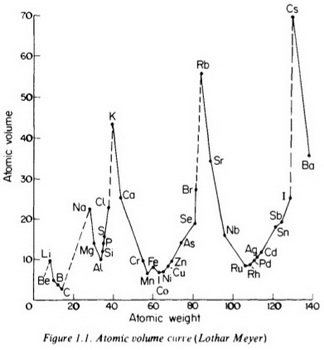
(from www.slideshare.net)
It showed a periodic pattern as the atomic volume rose to peaks and then fell again.
For example, the alkali metals occupied the peaks of the curve, which dropped to a minimum before rising to the next peak.

