Question #62413
1 Answer
Explanation:
I would have to say iodine monochloride,
The reactivity of diatomic halogens actually decreases as you move down group 17 of the periodict.
This happens because as you move down the group, the size of the atoms, and implicitly of the molecules, increases.
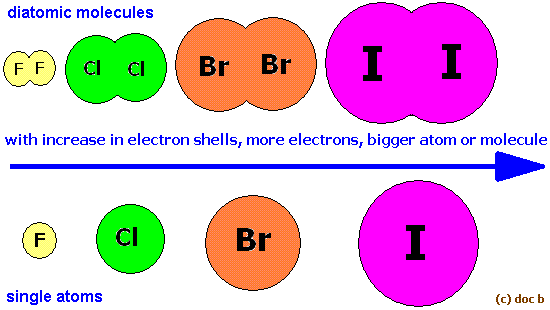
This means that the molecules have bigger electron clouds and will experience stronger London Dispersion Forces as you move down the group.
On the other hand, iodine monochloride is very reactive because of the difference in electronegativity between iodine and chlorine, which makes for a polar molecule.
The molecule will actually release

