Question #a465b
1 Answer
Explanation:
You're dealing with the oxidation of benzyl alcohol, which is the common name given to phnehymethanol,
The permanganate anion,
Now, the reaction will also produce water and aqueous potassium hydroxide, but these products are not important here. All you really need to know is that benzyl alcohol and benzoic acid have a
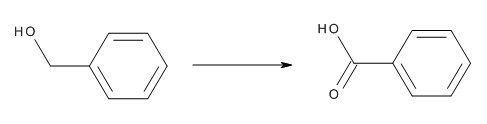
The reaction can be written as
#"C"_6"H"_5"CH"_2"OH"_text((aq]) stackrel(color(red)("KMnO"_4)color(white)(aaa))(->) "C"_6"H"_5"COOH"_text((aq])#
So, every mole of benzyl alcohol that takes part in the reaction will theoretically produce one mole of benzoic acid.
Use the molar mass of benzyl alcohol to determine how many moles you have in that
#1.05color(red)(cancel(color(black)("g"))) * ("1 mole benzyl alc.")/(108.14color(red)(cancel(color(black)("g")))) = "0.00971 moles benzyl alc."#
Theoretically, the reaction should produce
#0.00971 color(red)(cancel(color(black)("moles benzoic acd."))) * "122.12 g"/(1color(red)(cancel(color(black)("mole benzoic acd.")))) = "1.186 g benzoic acd."#
This is how much benzoic acid is produced by a reaction that has a
However, you know that you collected
Percent yield is defined as
#color(blue)(|bar(ul("% yield" = "what you actually get"/"what you should theoretically get" xx 100))|)#
Plug in your values to get
#"% yield" = (0.21 color(red)(cancel(color(black)("g"))))/(1.186color(red)(cancel(color(black)("g")))) xx 100 = "17.71%"#
You need to round this off to two sig figs, the number of sig figs you have for the mass of benzoic acid
#color(green)(|bar(ul(color(white)(a/a)"% yield" = 18%color(white)(a/a)))|)#

