Question #2c7a3
2 Answers
Caesium fluoride
Explanation:
The electronic structure of caesium is
The halogen in period 2 is fluorine.
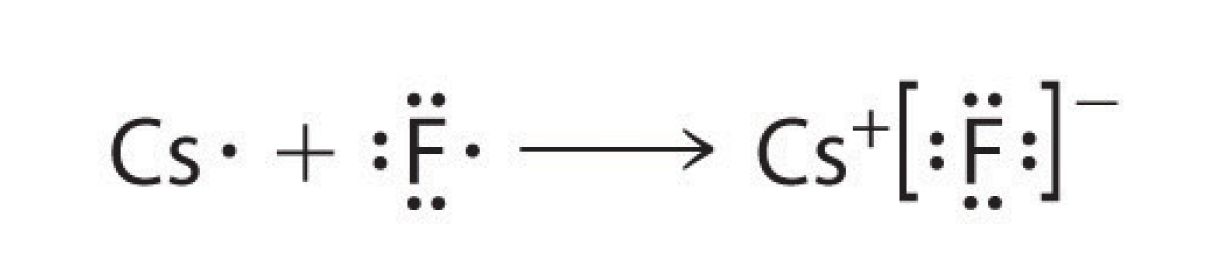
The reaction is:
Caesium fluoride is the binary compound formed.
Cesium fluoride,
Explanation:
This question can be answered by carefully inspecting the periodic table, so make sure that you have one near by.
So, the first important thing to notice here is that this reaction will produce an ionic compound. This is the case because the element located in group 1 is said to donate an electron to an element located in group 17, i.e. to a halogen.
Notice that the element located in group 1 donates a
A quick look in the periodic table will reveal that this element is cesium,
Since cesium donates one electron, it forms
This means that the chemical formula for this ionic compound is
To name the compound, start with the name of the cation, cesium. The anion will take the suffix -ide, which means that the name of the compound will be cesium fluoride.