Question #495b7
1 Answer
Explanation:
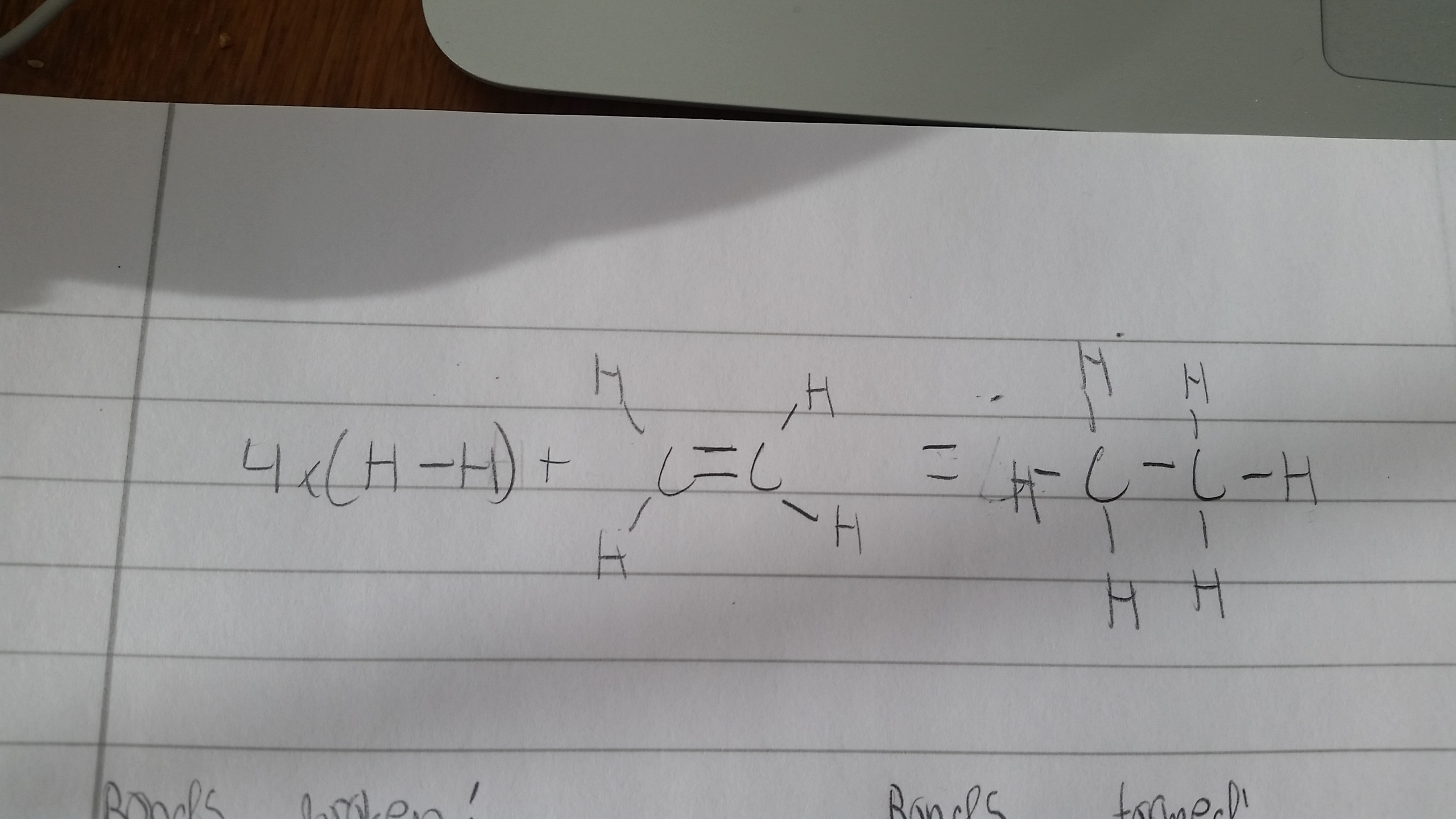
If the reaction in question is exactly as you have written the answer should be +2034 kj per mol. But when hydrogenating ethene generally the reaction is:
To calculate this we must use Bond energies (or Bond enthalpies). My values were taken from my course's data booklet and I advise to check your data booklet too to see that it corresponds.
The answer is found by comparing energies from "Breaking and making bonds".
Bonds broken= 4 H-H bonds, 4 C-H bonds, , 1 C-C double bond.
(as breaking is Endothermic, Delta H values are pos)
Bonds formed= 6 C-H bonds, 1 C-C bond
(As making is Exothermic, Delta H values are neg.)
Then the total enthalpy(energy) is:
or
This picture also shows the Lewis structure of all molecules involved.