Question #ca2e6
1 Answer
Generally solids which have free moving electrons, like the transition metals in the first d-block in the Periodic table.
Explanation:
The only criteria to be an electrical conductor is that you have free moving electrons or ions. When it comes to solids, the metals from the transition metals in the periodic table is a famous case.
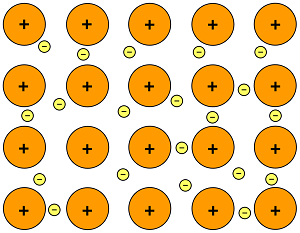
All of them have a so-called metallic bond. This type of bond is a large positive ion surrounded by a "sea" (or a large quantity) of electrons that move freely between all ions in the structure, meaning it can transfer electricity as the electrons are free to move and can pass a current. This is also why graphite can conduct electricity despite being a non-metal, it has free moving electrons in its structure.
Similarly, this is why salts like NaCl can transfer electricity when dissolved in water. When it's in a solid state it's ions are "locked in place" , but when they are dissolved in water the ions are free to move, and hence the solution can conduct electricity.
This diagram is a good illustration of a metallic bond: