What is the nuclear notation for a silicon atom that has #15# neutrons?
2 Answers
Well, you know it's silicon, so
Explanation:
If it's
Refer to the explanation.
Explanation:
Nuclear notation (nuclear symbol) is a shorthand way to represent the nucleus of an isotope of an element. The nucleus of an atom contains protons and neutrons. The sum of the protons and neutrons is the mass number.
Isotopes differ in the number of neutrons, and therefore their mass numbers. Nuclear notation contains the element's symbol, X, along with the mass number, A, or the atomic number, Z, or both A and Z.
The diagram below gives a general description about how to write the nuclear notation of an isotope of an element correctly.
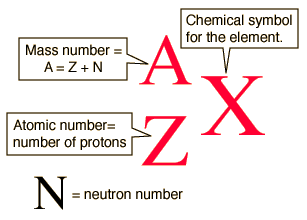
The diagram shows the correct way to write the nuclear notation for an isotope. First write the symbol (X) of the element. Then write the atomic number (Z) as a subscript on the left-hand side of the symbol, and the mass number (A) as a superscript on the left-hand side. Looking at the example, you can also see that the number of neutrons (N) can be determined by subtracting the atomic number, Z, from the mass number, A.
In your question, you are asked to determine the nuclear notation
for silicon-29, where A=29, and Z=14. For all silicon atoms, Z is always 14, because that is the atomic number of silicon, which is the number of protons in the nucleus of every silicon atom.
The nuclear notation of silicon-29 is:
Notice that the atomic number (14) is the subscript, and the mass number (29) is the superscript.
Compare the nuclear notation of silicon-29 to that of silicon-30.
Notice that only the mass number, A, has changed, but the atomic number, Z, is still 14.


