Under normal conditions, how could you reduce air pressure?
1 Answer
In an air-tight container, permit some of the water to drain away without letting air back in to take its place.
Explanation:
Here is series that might help. This assumes we have a container with an air-tight lid.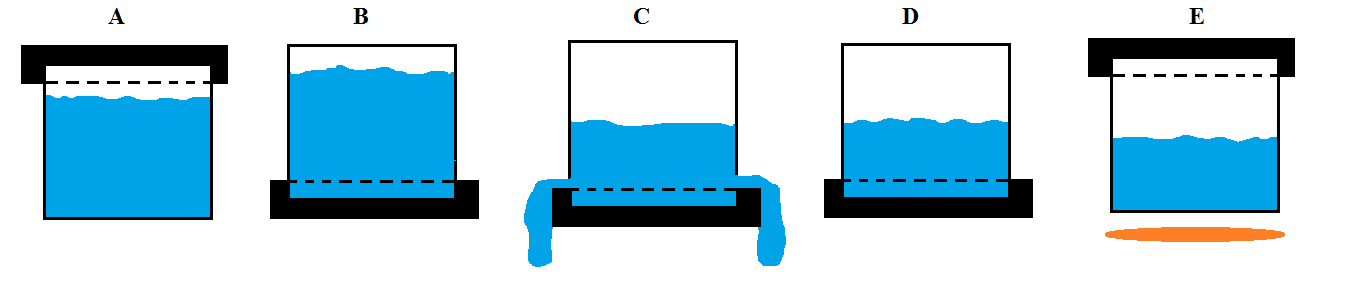
Step A
Fill air-tight container with water.
Step B
Invert the air-tight container.
Step C
Loosen the lid allowing some of the water to escape and creating a partial vacuum.
Step D
Re-tighten the lid before all the water has escaped.
Step E
Return the container to its upright condition without loosening the lid and apply heat.
Note: This will only be partially successful since once the water is heated the vapor will quickly restore the internal air pressure to and above normal.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Depending upon what is meant by "in normal conditions"
you might be able to take your pot of water to the top of a mountain to boil it. Under "normal conditions" the air pressure at the top of a mountain will be lower than at the bottom of the mountain.
