Question #12905
1 Answer
Feb 14, 2017
I'm not sure what you did, but I don’t think it was correct.
Explanation:
First, you prepared a standard curve, in which you plotted the absorbances of known concentrations of the drug.
I presume you got a graph something like this.
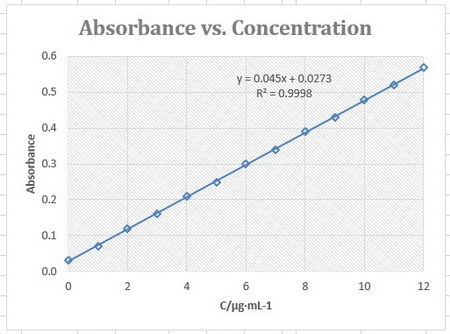
You use this plot to determine the concentration of your unknown.
Then you diluted your unknown sample by a factor of 1000 (?).
You measured its absorbance and got a value of 0.247.
From the regression equation,
The concentration of the drug in your diluted sample was 4.88 µg/mL.
Thus, the concentration in your original sample was 4.88 mg/mL or 48.8 mg/10 mL.

