How many, and of what type are the #sigma# and #pi-"bonds"# in #2-"methylphenol"#?
1 Answer
Jul 1, 2017
In
Explanation:
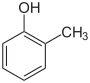
We gots isomeric
Now I count......
So that is
In

We gots isomeric
Now I count......
So that is