...Huh? 0 what?
The normal boiling point of water is #100.00^@ "C"#, and the normal freezing point of water is indisputably #0.00^@ "C"#. There is no need to discuss the so-called "vaporization point" because it's a synonym... (for what?)
The phase diagram here quite clearly proves it.
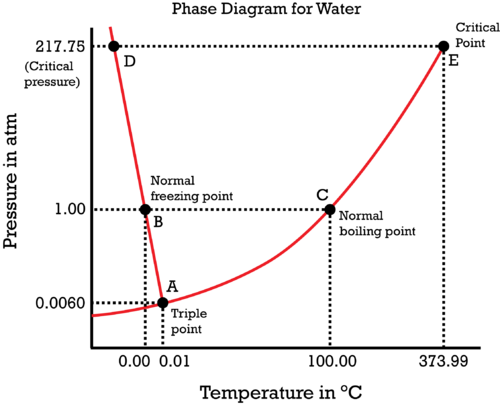
The "normal" phase transition temperatures by definition are the temperatures at #"1.00 atm"# where phase transitions will occur.
If you cross a phase coexistence curve (such as #bar(AD)# or #bar(AE)#), you transition to another phase. The easiest and most common way to do that is to heat or cool, i.e. move horizontally. Rightwards = heating up.
Hence, at #"1.00 atm"#, the normal freezing/melting point is at #0.00^@ "C"# and the normal boiling/condensation point is at #100.00^@ "C"#, as indicated by the dotted lines and, well, the words on the image.
QED
So, can you "prove" that the critical point is at #373.99^@ "C"# and #"217.75 atm"#? Can you convince yourself that the triple point is at #"0.0060 atm"# and #0.01^@ "C"#?


