Question #66680
1 Answer
Jan 7, 2018
It causes the ions to form ionic lattices
Explanation:
When an electron is transferred from one atom to another, the two atoms become charged and are referred to as ions. These positive and negative ions are strongly attracted to each other, forming a lattice as pictured.

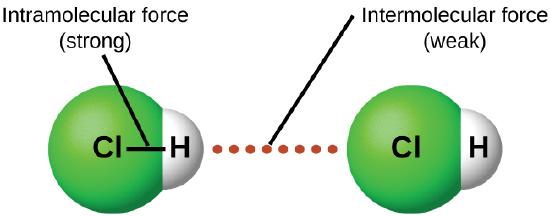
With covalent compounds, electrons are being shared between two atoms and have what are called intermolecular forces. These forces do not form lattices and are not nearly strong as ionic bonds.