Question #27e55
1 Answer
Oct 10, 2017
I'm afraid that more information is needed to answer the question. Here is just a brief example.
Explanation:
A half cell is a virtual concept to explain the principle of a chemical cell.
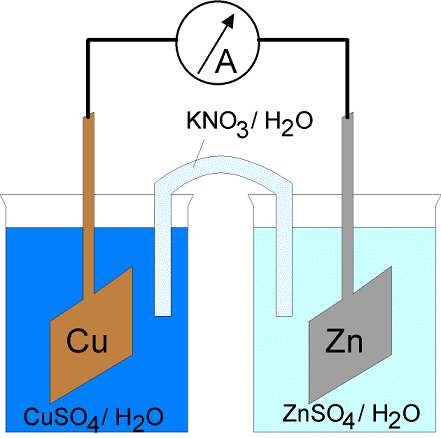
[Example] In a Daniel cell, you can think of the two isolated half cells.
(Anode)
(Cathode)
[How to connect?]
If you mean valancing the electron, just treat the equations like a simultaneous equation and erase
In the example shown above, just add (1) and (2) and you acquire
If you mean connecting the half cell physically, you need salt bridge. See the figure cited from https://en.wikipedia.org/wiki/Salt_bridge
**