Question #2d8b3
1 Answer
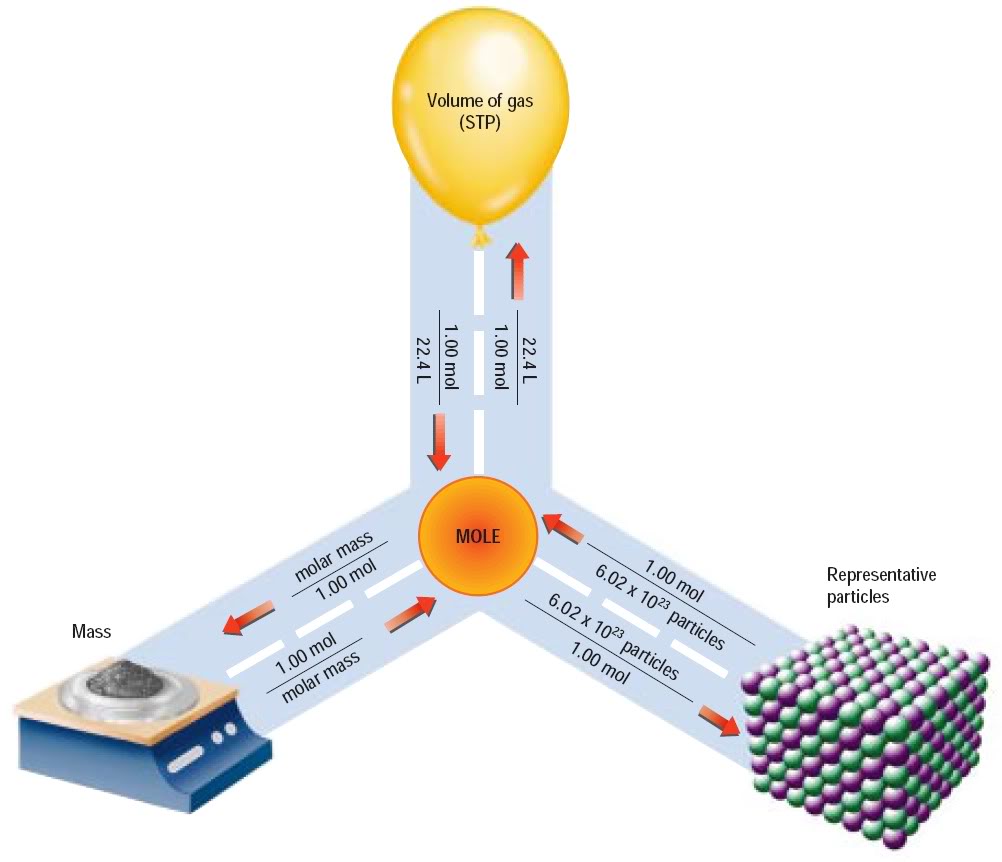
Convert your mass to moles, extract the molar percentage of the element of interest, and then convert those moles to atoms.
Explanation:
The first one needs to have the molecular weight
TOTAL =
The second part needs Avogadro's Number (units/mole),
Followed by the percentage of the element of interest (C) in the whole.
EXAMPLE Worked Problem:
How many atoms of oxygen are in 3.9g of methanol (
Molecular weight of methanol:
TOTAL =

