Question #7eca9
1 Answer
You may work it out from the "size" of each atom's nucleus and the electrons around it, or look it up in a table.
Explanation:
We know that the atoms generally decrease in size as they move left-to-right in a row of the Periodic Table of the Elements, and increase as they move down in a column, or Group. There is a sharp reversal going from the last element in a row to the first one of the next row.
Once the initial atomic sized are determined, we can observe that adding electrons (- charges) will increase the effective size, and removing them (+ charges) will decrease the effective size.
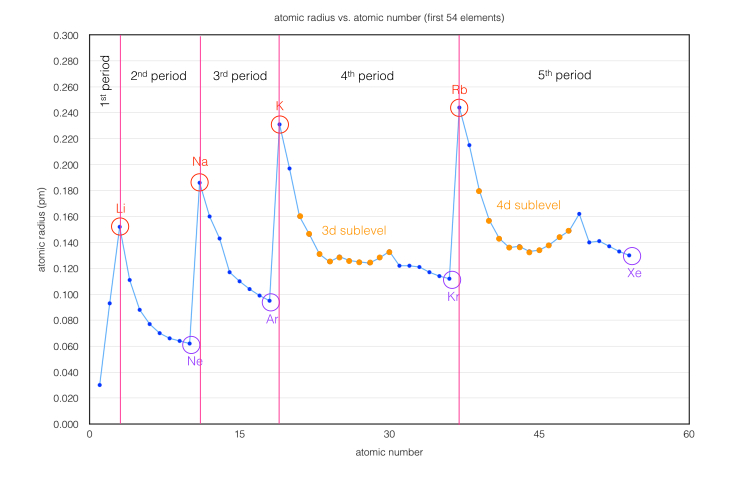
Given:
"Normal" sequence (decreasing):
First Row:
Second Row:
Ionic changes in each row:
So, relatively,
Considering the effect of charge balance, more electrons relative to protons (-) expand the radius, and fewer electrons (+) decrease the effective radius.
Thus, "isoelectronic" atoms can be compared:
Thus, the final order of the relative size of the ions listed is:
Actual values (picometer):
http://abulafia.mt.ic.ac.uk/shannon/ptable.php

