What are #"metallic properties"#, and how do these arise on account of #"metallic bonding"#?
2 Answers
So what are the properties of metals?
Explanation:
I can think of 4 defining characteristics:
And metallic bonding is typically described as
Metals atoms within the lattice can thus move with respect to each other, WITHOUT disrupting the electrostatic bonding interaction, the which is based on free electrons that can move across the entire lattice (and transfer heat or charge!). Metals also typically exhibit metallic lustre, for which the delocalized electrons are also held to be responsible.
Using metallic bonding we can explain many characteristics of transitional elements.
MELTING & BOILING POINTS
The transitional metals have high M.P & B.P.
Their M.P rise to maximum and then fall as the atomic number
Strong metallic bonds{M.B} between the atoms of these elements are responsible for higher M.P and B,P.
As unpaired electrons increases the M.B in that atom
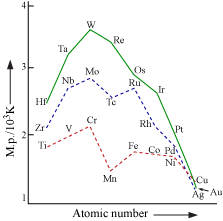
As we know the amount of unpaired electrons
You should remember that " Greater the no. of unpaired electrons greater the Metallic bonding".
{
HARDNESS & OTHER PHYSICAL PROPERTIES
As unpaired electrons
for eg: Cr, Mo ,and W have maximum no, of unpaired d electrons and therefore are hard metals whereas Zn ,Cu ,and Hg are not very hard metals due to absence of unpaired electrons and thus weak M.B.
hope it helps.


