Question #52fc8
1 Answer
Jan 17, 2018
There can be a lot of differences, but I can only think of some.
Explanation:
The first difference is their charges.
A proton carries a
An electron carries a
A neutron carries no charge.
Another difference is their mass.
As you can see here, an electron has approximately
Source: http://physics.bu.edu/~duffy/sc546_notes10/mass_defect.html
Next, the orbits and the positions of the particles.
Protons and neutrons are found in the nucleus of the atom, while electrons orbit the atom in their electron shells, which also include orbitals.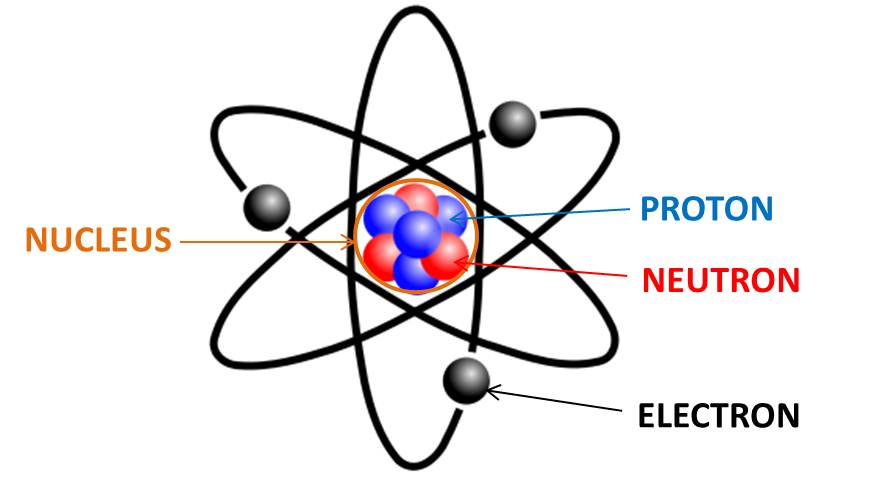
These are some examples I can think of.

