#20*cm^3# volumes EACH of carbon monoxide and oxygen gas are combined and combusted. What volume of gas remains AFTER the reaction?
2 Answers
There should be
Explanation:
Why? Well, take a captain at the stoichiometric equation....
Now it is a fact that equal volumes of gases contain the same number of molecules... The stoichiometric equation thus requires that
30 cm³
Explanation:
Carbon monoxide (CO) reacts with oxygen to form carbon dioxide: every 2 molecules of carbon monoxide react with one molecule of dioxygen (O₂) to form 2 molecules of carbon dioxide (CO₂).
The law of volumes of combining gases by Gay-Lussac mirrors the mole ratio:
2 CO + 1 O₂ → 2 CO₂ (moles);
2 + 1 → 2 (volumes).
So, for 20 volumes of CO (20 cm³ in the problem), only 10 volumes of O₂ at S.T.P. can react to yield 20 volumes of CO₂. The exceeding unreacted 10 cm³ of O₂, summed up to the 20 cm³ of CO₂, give rise to a total volume of 30 cm³ at S.T.P.
The following pictures show a schematic view (before & after lines) and a "realistic" view (third line).
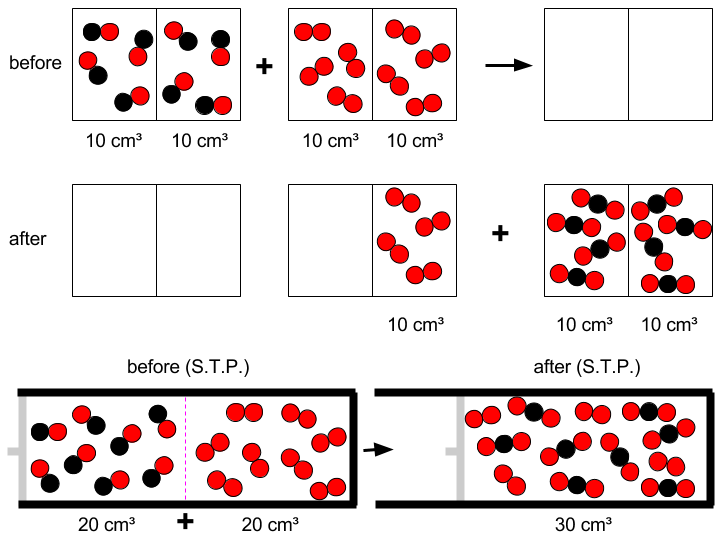
(Oxygen atoms are represented by red circles, while carbon atoms are black.)
This answer was elaborated by a student of mine, Alexia.
We hope it helps.


