A gas occupies a volume of 0.140 L at 35.0°C and 97.0 kPa. What is the volume of the gas at STP?
1 Answer
Sep 17, 2016
The volume of the gas at STP of 273.15 K and 100 kPa is 0.120 L.
Explanation:
STP currently has the values of 273.15 K and 100 kPa.
This is a question involving the combined gas law as shown below.
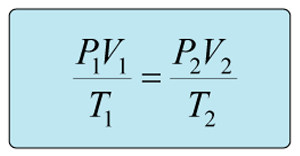
Rearrange the combined gas law to isolate

