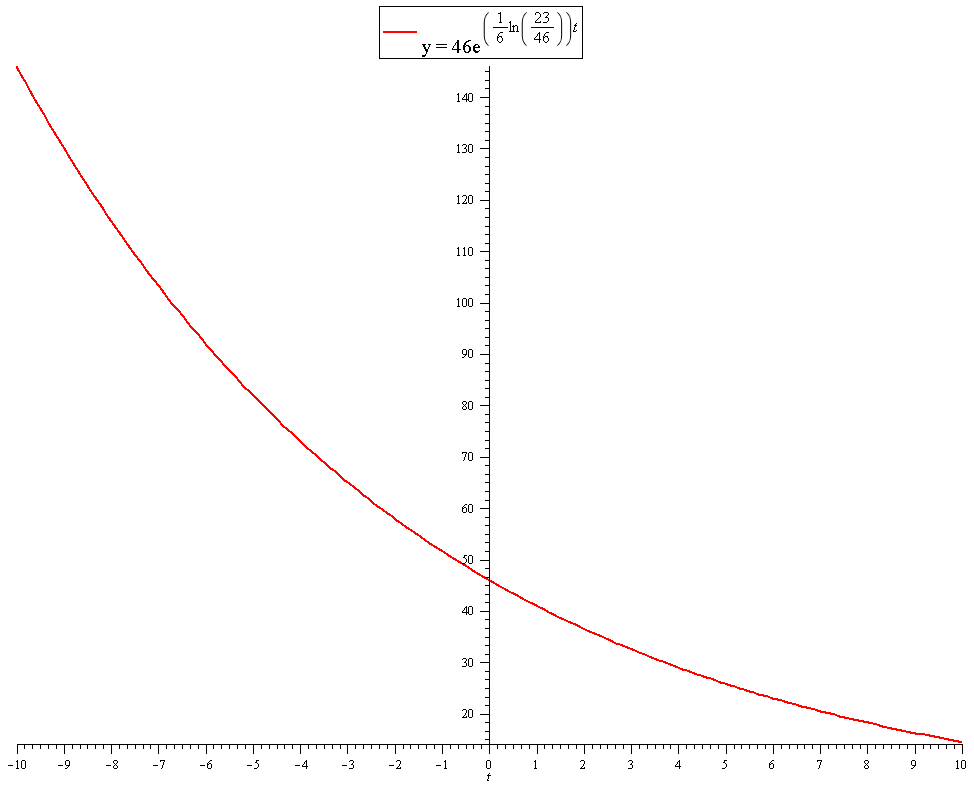
A serving of a popular cola soft drink has 46 mg of caffeine. Suppose the half-life for caffeine remaining in he body of a typical adult is 6 h. How do you write an exponential decay function that gives the amount of caffeine in the body after t hours?
1 Answer
Nov 27, 2017
See below.
Explanation:
We need an equation of the form:
Where
To find the the constant
We know the half life is 6 hours, so after 6 hours the amount
So:
Solve for
Taking natural logs of both sides:
So our equation is:
This could also be written:
TEST:
Graph of