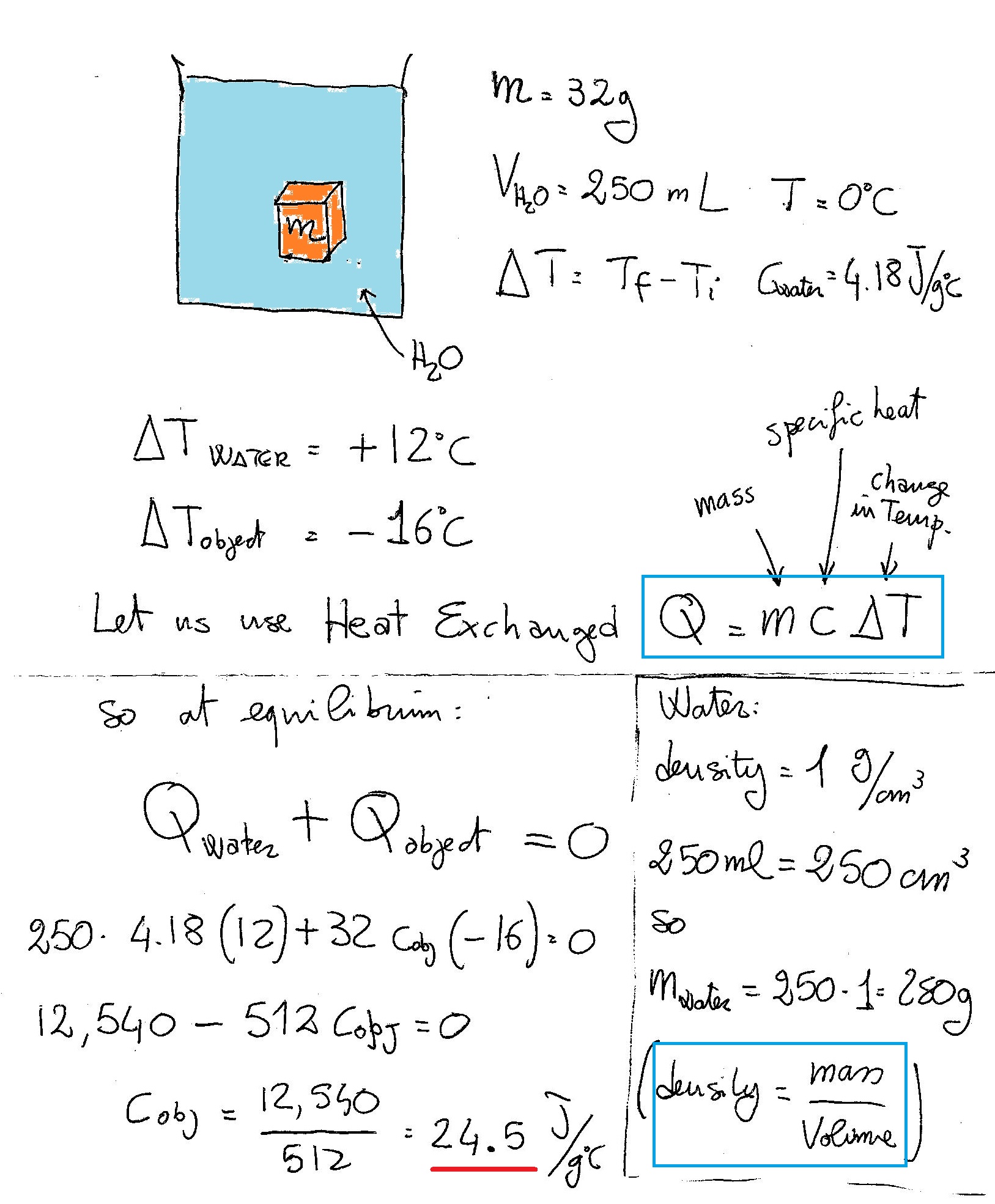
An object with a mass of #32 g# is dropped into #250 mL# of water at #0^@C#. If the object cools by #16 ^@C# and the water warms by #12 ^@C#, what is the specific heat of the material that the object is made of?
1 Answer
Nov 16, 2016
I found:
Explanation:
Have a look: