...............A very useful idea that is used to rationalize the formulae of organic compounds is the "degree of unsaturation". An alkane is fully "saturated", and it contains the MAXIMUM ALLOWABLE number of C-H bonds.
Alkanes are "FULLY saturated" and have a general formula of C_nH_(2n+2). Try this out for "methane," "ethane," ........."pentane, etc."
Each double bond, each olefinic bond or carbonyl group, OR a ring junction, corresponds to 1""^@ of unsaturation; i.e. 2 hydrogens LESS than the saturated formula. So according to the scheme, "ethane" has the saturated formula of H_3C-CH_3, but "ethylene", H_2C=CH_2, and "acetaldehyde", H_3C-C(=O)H has 1^@ of unsaturation. Halogen atoms count for one hydrogen; for nitrogen atoms, substract NH from the formula before assessing unsaturation; i.e. for "ethylamine," H_2NCH_2CH_3 rarr C_2H_6, i.e. "no degrees of unsaturation".
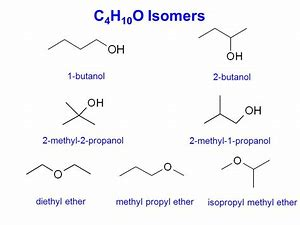
Given the former, a formula of C_4H_10O, the which is SATURATED, specifies FOUR alcoholic isomers, one of which can generate stereoisomers. And there are also THREE ethereal isomers...and some punter has kindly given the structures on the web..