#color(blue)("Step 1: Assume 100 g sample to find mass of each element")#
#"C" = 39.56" g"#
#"H" = 7.74" g"#
#"O" = 52.70" g"#
#color(white)(aaaa)#
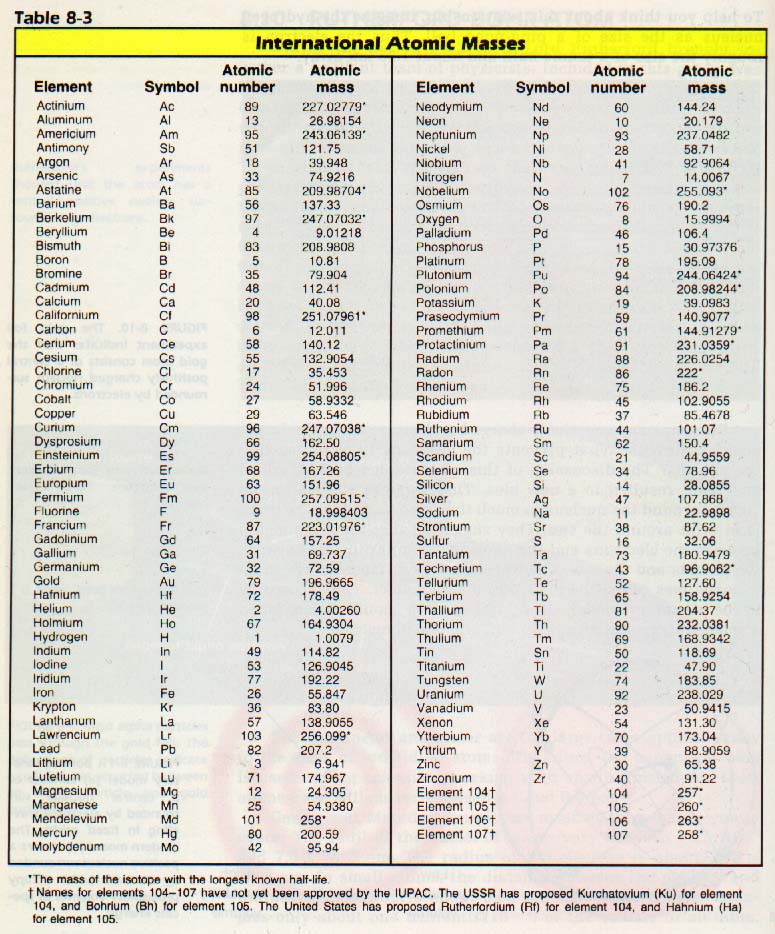
#color(blue)("Step 2: Find number of moles from the mass of each element.")# Use the periodic table to find molar masses of each element
#"C" = (39.56 cancel"g")/1 *(1" mol")/(12 cancel"g") = 3.29" mol"#
#"H" = (7.74 cancel"g")/1 *(1" mol")/(1.00 cancel"g") = 7.74" mol"#
#"O" = (52.70 cancel"g")/1 *(1" mol")/(16 cancel"g") = 3.29" mol"#
#color(white)(aaaa)#
#color(blue)("Step 3: Divide all the mole numbers by the smallest mole value of them all")#
#"C" = (3.29cancel"mol")/(3.29 cancel"mol") = 1.00#
#"H" = (7.74cancel"mol")/(3.29 cancel"mol") = 2.35#
#"O" = (3.29 cancel"mol")/(3.29 cancel"mol") = 1.00#
#color(white)(aaaa)#
#color(blue)("Step 4: Multiply the numbers by a factor which will give out a whole number ratio.")#
#"C" = 1.00 * (3) -> 3#
#"H" = 2.35 * (3) -> 7.05~~7#
#"O" = 1.00 * (3) -> 3#
#color(white)(aaaa)#
#color(blue)("Step 5: Combine elements and attach corresponding mole values."#
#color(white)(---)color(blue)("Then, find empirical formula mass using the periodic table"#
#color(white)(----)stackrel"empirical formula"||ul(C_(3)H_(7)O_(3))|| -> stackrel"empirical formula mass"||ul((91" g")/"mol" )||#
#color(white)(aaaa)#
#color(blue)("Step 6: Divide given molecular formula mass by empirical formula mass")#
#color(white)(---)color(blue)("to get a factor.")#
#color(white)(aaaaaaaaa)("Molecular formula mass")/("Empirical formula mass") ->[(182 cancel"g")/(cancel"mol")]/[(91 cancel"g")/(cancel"mol")]= 2#
#color(white)(aaaa)#
#color(blue)("Step 7: Take this factor and multiply it to the empirical formula")#
#color(white)(---)color(blue)"to get the molecular formula of sorbitol"#
#color(white)(-----)stackrel"empirical formula"||ul(C_(3)H_(7)O_(3))||xx2 -> stackrel"molecular formula"||ul(color(orange)(C_(6)H_(14)O_(6)))||#