Compare the structures and functions of the biomolecules lipids and starches?
1 Answer
WARNING! —This is a long answer—
Explanation:
STARCH
Structure
Starch consists of a large number of glucose units joined together.

There are two forms of starch.
Amylose is a linear chain of α-D-glucose units joined by 1→4 linkages.
Plant amylopectin has a branched structure in which about one residue in every twenty is also has 1→6 linkages.
The body stores glucose as glycogen in muscle and liver. The structure of glycogen is like that of amylopectin, but the 1→6 branches occur about every 8 to 10 glucose units.
Starch and glycogen contain many OH groups. They are hydrophilic molecules.
Function
Starch is the main source of energy in plants.
Glycogen is the main source of energy in the body. Muscle glycogen is used in the synthesis of adenosine triphosphate (ATP) during muscle contraction. Liver glycogen maintains the blood glucose concentration.
LIPIDS
Lipids have many different structures and functions. They are insoluble in water. Many have a polar "head" and a nonpolar "tail". The most common types of animal lipids are fats, phospholipids, steroids.
FATS
Structure
Fats are esters of three fatty acids and glycerol (triglycerides).

Function
Fats store energy, help to insulate the body, and cushion and protect organs.
PHOSPHOLIPIDS
Structure
A phospholipid consists of two fatty acids, a glycerol unit, a phosphate group. and a polar molecule. The phosphate group and polar head region of the molecule are hydrophilic. The fatty acid tail is hydrophobic.

Function
In water, phospholipids orient themselves into a bilayer. The nonpolar tail faces the inner area of the bilayer. The polar head faces outward and interacts with the water.
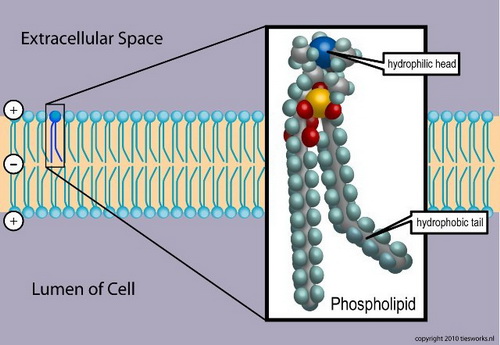
Phospholipids are a major component of the cell membranes.
STEROIDS
Structure
Steroids have a carbon backbone that consists of four fused ring-like structures. A common steroid is cholesterol.

Function
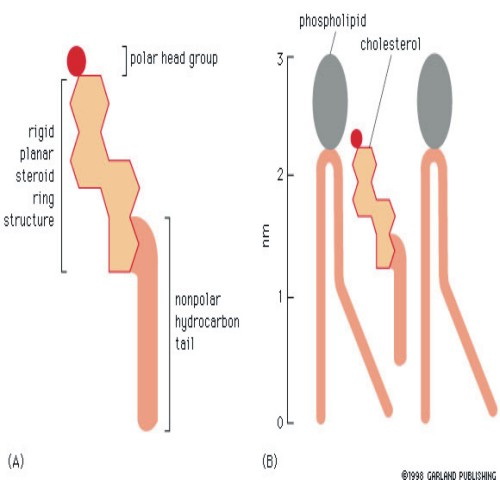
Cholesterol has a polar head and a nonpolar tail. It fits well between the phospholipids in the cell membrane. It maintains the fluidity of the cell membrane.

