Discuss why benzene is a good solvent (even though it is non-polar). Additionally, why is the dipole moment not exactly zero?
1 Answer
Well, a lot of nonpolar solutes (e.g. diethyl ether, small alcohols, acetone, etc) exist in organic chemistry, so benzene CAN be good solvent.
Otherwise, perhaps the best reason why it is still a good (nonpolar) solvent is that it is very unreactive. It is stabilized since its
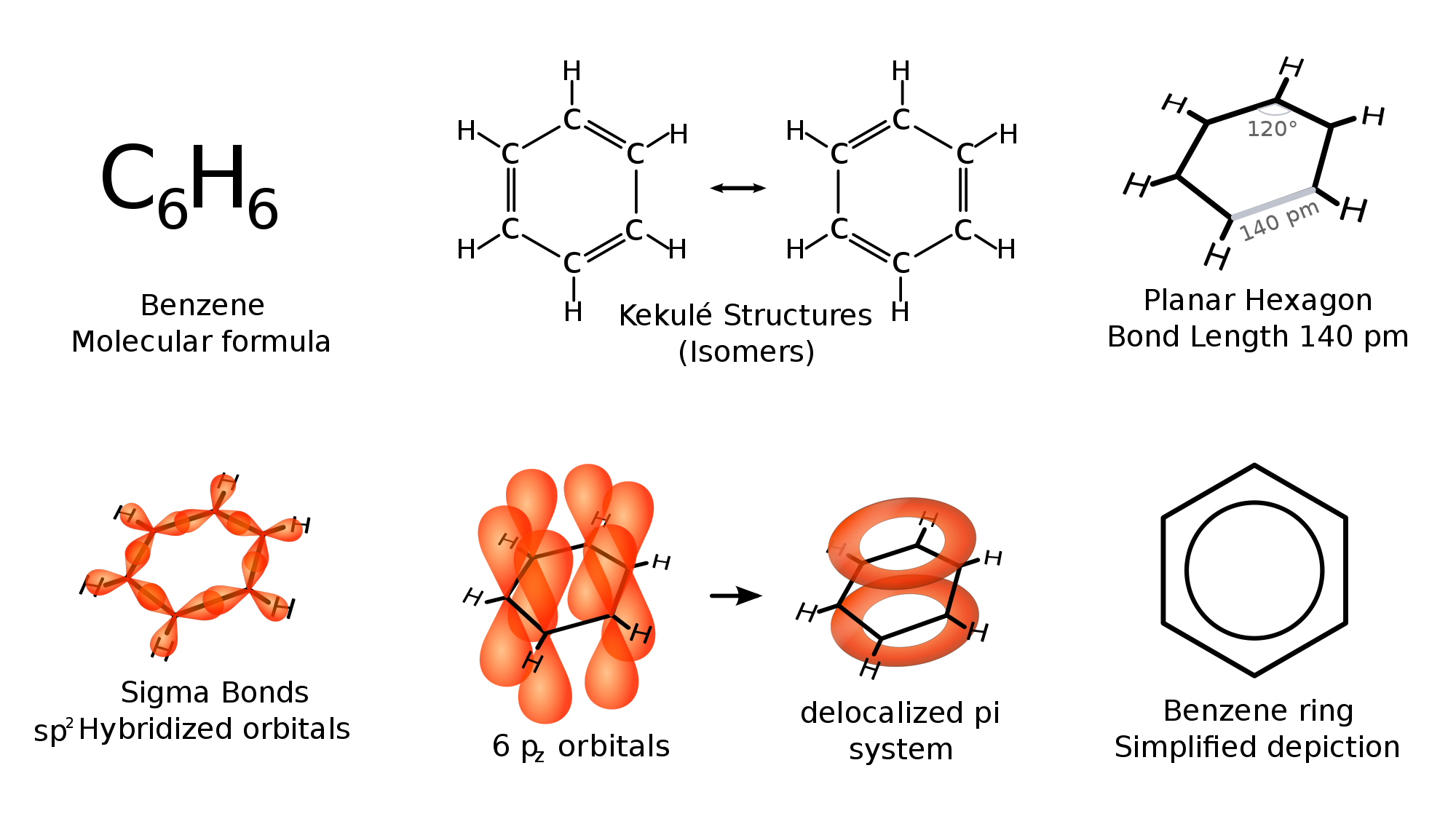
The last thing you want is for your solvent to react with your reactants.
As for the dipole moment, for MOST intents and purposes, it is zero.
If we want to get technical, we would NOT assume that the motions of the hydrogen and carbon nuclei is slow compared to the electrons (meaning, we would NOT assume the Born-Oppenheimer approximation applies).
In that case, like methane (mentioned in Group Theory and Chemistry by Bishop) the dipole moment of benzene would be on the order of
This is because the nuclei shuffle around semi-randomly, not necessarily in the same direction and not necessarily in-phase. The symmetry of the molecule is then broken slightly, and a slight dipole moment is produced.

