For a Le Chatelier Principle lab, I added Thymol Blue (blue) and HCl together. What would be the equilibrium equation to represent this situation?
The equilibrium equation written on the lab is:
Thymol Blue (blue) + #H^+# #rightleftharpoons# Thymol Blue (yellow)
How would I adjust this equilibrium equation to show that I added HCl?
Thank you in advance!
The equilibrium equation written on the lab is:
Thymol Blue (blue) +
How would I adjust this equilibrium equation to show that I added HCl?
Thank you in advance!
1 Answer
Here's what I got.
Explanation:
The equation itself does not change, but you should change the equilibrium sign to show that the forward reaction becomes favored.
The equilibrium equation given to you
#color(blue)("thymol b ") + " H"_ ((aq))^(+) rightleftharpoons color(gold)("thymol b")#
shows you what happens when you add hydrochloric acid, a strong acid represented in solution by
As you know, equilibrium reactions are governed by Le Chatelier's Principle, which states that the system will shift in such a way as to reduce a stress placed on its position.
Now, when you adding hydrochloric acid, i.e. increasing the concentration of hydrogen ions present in solution, the system must shift in such a way as to decrease the concentration of hydrogen ions.
Since the hydrogen ions are present on the reactants' side of the equation, decreasing their concentration means shifting the equilibrium to the right.
The forward reaction
#color(blue)("thymol b ") + " H"_ ((aq))^(+) -> color(gold)("thymol b")#
consumes hydrogen ions and molecules of thymol blue in the blue form and produces molecules of thymol blue in the yellow form, as you can see in the image below in the bottom equilibrium reaction.
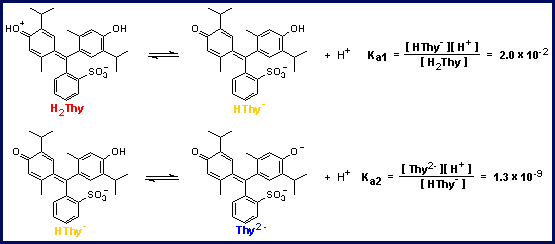
In this regard, the equilibrium symbol should be changed to look like this
#stackrel(color(white)(aaaaaa))(->)#
#larr#
This shows that the forward reaction, which is represented by the right arrow, is now being favored over the reverse reaction, which is represented by the left arrow.
Notice that if you continue to increase the concentration of hydrogen ions, i.e. decrease the pH of the solution, you get this
#color(gold)("thymol b ") + " H"_ ((aq))^(+) -> color(red)("thymol b")#
Thymol blue solutions are blue in color at


