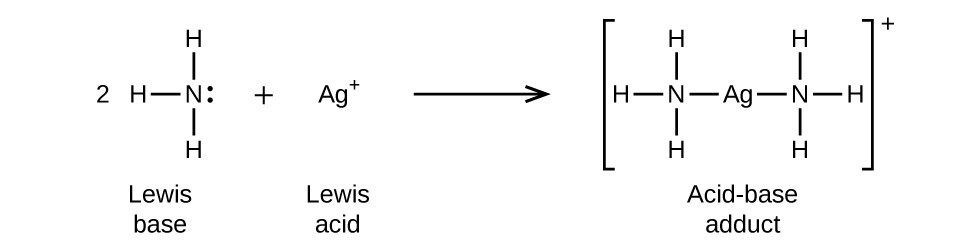
Given, 2NH3 + Ag+ Ag(NH3)2+, what is the lewis acid and the lewis base in this equation?
1 Answer
Dec 21, 2015
You meant to write:
#2"NH"_3(aq) + "Ag"^(+)(aq) -> ["Ag"("NH"_3)_2]^(+)(aq)#
This is a complexation reaction, so ammonia is interacting with the silver cation, but isn't contributing to the overall charge.
Since we would see ammonia associating itself around silver and not the other way around, silver thus accepts electrons. So, silver is then the lewis acid.
Can you see why ammonia is the lewis base, then?