Given the reaction: #HC_2H_3O_2(aq) + H_2O rightleftharpoons H_3O^+(aq) + C_2H_3O_2^(-)(aq)# When the reaction reaches a state of equilibrium, what happens to the concentrations of the reactants?
1 Answer
For your reaction in particular, we would have that the concentrations reach values that satisfy
#K_a = (["H"_3"O"^(+)]["C"_2"H"_3"O"_2^(-)])/(["HC"_2"H"_3"O"_2]) = 1.76 xx 10^(-5)# .
The reaction quotient
In general, at dynamic equilibrium, the concentrations of the reactants and products change in such a way that the concentrations stay constant. The rate of the forwards and backwards reactions become the same, and thus the rate of forming products equals the rate of forming reactants.
The products still get made in the forward reaction and the reactants still get made in the reverse reaction; there is just no observable change, and we say that the reaction is in a steady state.
For example, in this reaction of
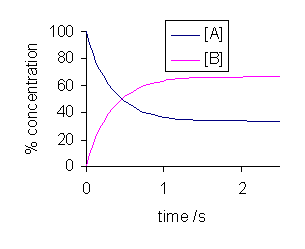
In other words, ongoing processes occur to change the concentrations, but the forward and reverse directions cancel out and no changes are observed, even though they occur.

