How can chirality influences or forms different conformations in case of vinyl polymers (‐CH2‐C*HR‐)n ?
1 Answer
Feb 8, 2018
The classic example is polypropylene....
Explanation:
Polypropylene can be prepared with 3 different stereochemistries....
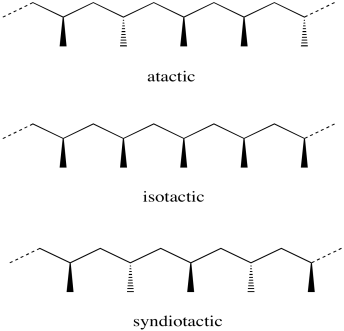
These polymers are prepared by catalysts, i.e. metal centres with specialty ligands, that DIRECTLY influence the geometry of the polymer chain. In the best circumstances the reaction can be made to be stereospecific, i.e. the given geometry of the catalyst gives rise to a definite stereochemistry in the resultant polymer.

