How can I draw the Lewis structure for PH3?
1 Answer
Jul 16, 2014
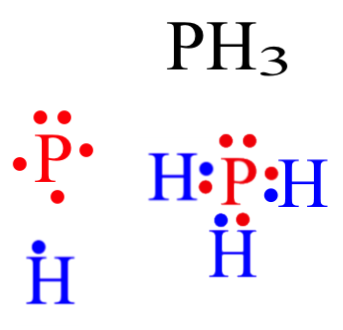
Phosphosus has an electron configuration of
Therefore, it has a dot digram in the red of two electrons on top and one electron on each side. Providing for three open bonding sites.
Hydrogen has an electron configuration of
Each hydrogen now has two electrons to complete the rule of duet.
I hope this was helpful.
SMARTERTEACHER

